Separating Benzoic Acid, 4-choloraniline and cholesterol using extraction
Written by Brandon Ritter
What does it mean to “work up” an organic reaction?
Purpose: The purpose of this lab was to separate benzoic acid, 4-choloraniline and cholesterol using extraction.
Reaction Equations: Missing 🙁
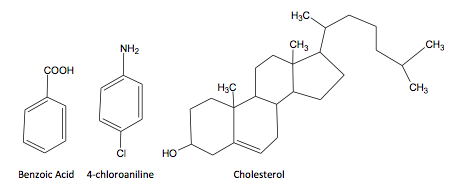
Structures:
Calculations: Missing 🙁
Results:
Benzoic Acid:
- Melting Point: 121.9-123.1⁰C
- Mass: 0.369g
4-chloroaniline:
- Melting Point: 68.6-70.2⁰C
- Mass: 0.043g
Cholesterol:
- Melting Point: 146.7-147.2⁰C
- Mass: 0.205g
Discussion:
- Our percent recovery for benzoic acid was really good, we got a lot more recovered benzoic acid product than expected. We got slightly less cholesterol product than we expected but we still got a decent amount. We were not able to recover a lot of solid 4-chloroaniline at all, but luckily we were able to recover enough to get the needed melting point data.
- All of our melting point ranges were within a very close range of the actual melting points. This tells us that the products were recovered are pretty pure.
- Our melting points are pretty close so there is no evidence of error to show our compounds are not pure.
- Based on our experimental results we can come to the conclusion that extraction is a fairly good method for recovering solid products.
Conclusion:
Based on our experimental results we can conclude that extraction is a good method of obtaining the desired isolated products from a mixed unknown starting material. We obtained 0.369g of benzoic acid, 0.043g 4-chloroaniline, and 0.205g cholesterol. Our melting point ranges that we obtained for our experimentally obtained solid products were really close to the actual ranges for the solids we isolated, thus telling us that our isolated solids were relatively pure.