The Diels-Alder Reaction
Understanding Diels-Alder Stereochemistry: Key Reactions and Products
The Diels-Alder reaction between cyclopentadiene and maleic anhydride — documented in the student report below — is one of the most studied [4+2] cycloadditions in organic chemistry. Before examining that experiment, this section addresses several closely related reactions that build on the same mechanistic principles and are frequently studied in advanced coursework.
5-Fluorocyclopentadiene + Maleic Anhydride: Major Product and Stereochemistry
When 5-fluoro-1,3-cyclopentadiene reacts with maleic anhydride, the fluorine substituent at C-5 breaks the facial symmetry of the diene, making the two faces of the molecule non-equivalent. The major product is the syn-endo adduct, in which maleic anhydride approaches from the same face as the fluorine substituent (syn addition) under endo transition state geometry. This gives the product the stereodescriptor (3aR,4R,7S,7aS,8S) for the major isomer, with the fluorine substituent in the syn-7 position of the resulting bicyclic framework.
This facial selectivity is explained by the Houk model, which predicts that electron-withdrawing substituents at C-5 direct dienophile approach to the syn face due to hyperconjugative and electrostatic stabilization of the transition state. This has been confirmed experimentally by Mcclinton and Sik, whose work on polyfluorocyclopentadienes established the stereochemical outcomes for this class of reactions. The syn/anti orientation of the fluorine relative to the anhydride bridge can be confirmed by NOE correlations between H-8 (the fluorine-bearing carbon) and the bridgehead protons H-3a and H-7a.
1,2,3,4-Tetramethylcyclopentadiene + Maleic Anhydride: NOESY Analysis
The reaction of 1,2,3,4-tetramethyl-1,3-cyclopentadiene with maleic anhydride produces two diastereomeric adducts with molecular formula C₁₃H₁₆O₃ — an endo (major) and exo (minor) product. The four methyl groups create steric bulk that influences the product ratio, but the endo product remains kinetically favored under standard conditions consistent with the endo rule.
Distinguishing the major from minor product requires 2D NOESY NMR spectroscopy. The key diagnostic cross-peaks are:
- Endo product (major): NOESY cross-peaks are observed between the anhydride CH protons (H-5 and H-6) and the methyl groups on the diene-derived ring, confirming the anhydride bridge is positioned over the methyl-substituted ring in the endo configuration. A characteristic singlet near 1 ppm integrating for 6H (two equivalent methyl groups) is also diagnostic.
- Exo product (minor): The anhydride CH protons do not show cross-peaks to the methyl groups, as the anhydride bridge points away from the substituted ring in the exo configuration.
The ¹H NMR spectrum of the major product shows two peaks in the 13C spectrum near 137 ppm (alkene carbons) and 165 ppm (carbonyl carbons) of the anhydride, consistent with a single symmetric anhydride environment.
Cyclooctatetraene + Maleic Anhydride: Multi-Step Sequence
A three-step sequence commonly encountered in advanced coursework begins with cyclooctatetraene (COT) reacting with maleic anhydride to give the initial Diels-Alder adduct (Reppe’s anhydride). This adduct is then esterified with methanol and sulfuric acid to give the dimethyl ester, which undergoes a second Diels-Alder reaction with cyclopentadiene. The major isomer of the final product results from endo approach of cyclopentadiene to the less hindered face of the dimethyl ester dienophile. The stereochemistry of the final tricyclic product is determined by the facial selectivity of each cycloaddition step in sequence.
The Classic Reaction: Cyclopentadiene and Maleic Anhydride
The student experiment below examines the simplest and most foundational version of this reaction — unsubstituted cyclopentadiene reacting with maleic anhydride under mild conditions. Because no substituent is present to introduce facial selectivity, the stereochemical outcome is governed entirely by the endo rule: the kinetic product is the endo isomer, cis-norbornene-5,6-endo-dicarboxylic anhydride, formed when the carbonyl groups of maleic anhydride orient toward the diene in the transition state. The endo product is confirmed by melting point, ¹H NMR coupling patterns, and IR spectroscopy as described in the report below.
The Diels-Alder Reaction
By: Kayla Powers and Jakkrit Suriboot
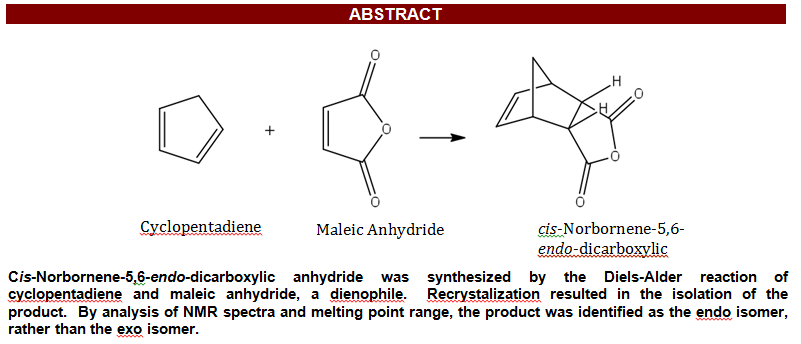
Introduction Diels-Alder reactions are used for synthesizing new carbon-carbon bonds and more specifically, six-membered cyclic compounds. In addition, this reaction synthesizes compounds that are otherwise difficult to obtain, such as bridged bicyclic compounds. A key characteristic of these reactions is their stereospecificity. Based on the interaction between a conjugated diene and a dienophile, different stereoisomeric compounds are formed. The Diels-Alder reaction is categorized as a pericyclic reaction, which involves the overlap of spatial orbitals as well as the hybridization and delocalization of the molecules.1 As a unique characteristic, this reaction is characterized as a concerted cycloaddition reaction indicating a lack of intermediate in the mechanism.
Stereochemistry represents a major component of the Diels-Alder reaction. Due to the interaction and arrangement of a cyclic diene and a dienophile, an endo and exo product can be formed characterizing the reaction as stereo- and regioselective. By analysis of NMR spectroscopy and physical properties of the specific isomers, the difference between the possible products can be identified.
Interesting products of the Diels-Alder reaction are cyclic compounds with chlorine-containing substituents that act as powerful insecticides. Insecticides have been commonly used to treat pests in various types of fruits, vegetables, and crops. Because of the negative affect on the environment, certain pesticides have remained unused and alternative methods involving the elimination of pests have been investigated. Strategies, such as using hormones have been explored with haste because of the potential damage many pests have on agricultural produce.2 These compounds have been researched and related back to their concerted cycloaddition mechanism.
Reaction Mechanism The scheme below depicts the concerted mechanism of the Diels-Alder reaction of cyclopentadiene and maleic anhydride to form cis-Norbornene-5,6-endo-dicarboxylic anhydride.
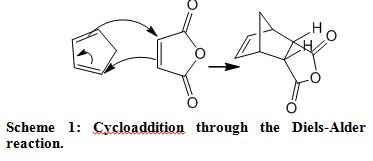
Results and Discussion When combining the reagents, a cloudy mixture was produced and problems arose in the attempt to completely dissolve the mixture. After heating for about 10 minutes and magnetically stirring, tiny solids still remained. The undissolved solids were removed form the hot solution by filtration and once they cooled, white crystals began to form. Regarding the specific reaction between cyclopentadiene and maleic anhydride, the endo isomer, the kinetic product, was formed because the experiment was directed under mild conditions. The exo isomer is the thermodynamic product because it is more stable.3
A total of 0.47 g of the product was collected; a yield of 27.6%. The melting point was in the range of 163-164 °C which indicates the absence of impurities because the known melting point of the product is 164 °C.
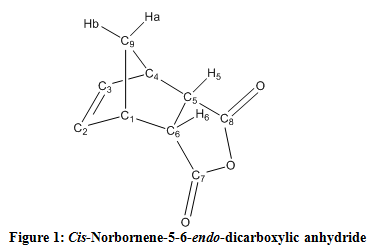
The 1H NMR spectrum of the product revealed a peak in the alkene range at 6.30 ppm, H-2 and H-3 (Figure 1). In addition, it exhibited two peaks at 3.57 and 3.45 ppm because of the proximity of H-1, H-4, H-5, and H-6 to an electronegative atom, oxygen. Finally, two peaks at 1.78 and 1.59 ppm corresponded to the sp3 hydrogens, Hb and Ha, respectively. Impurities that appeared included ethyl acetate at 4.03, 2.03, and 1.31 ppm as well as acetone at 2.16 ppm.
Regarding the 13C NMR, a peak appeared at 171.3 ppm, accounting for the presence of two carbonyl functional groups, represented by C-7 and C-8 in Figure 1. The alkene carbons, C-2 and C-3, exhibited a peak at 135.5 ppm, while the sp3 carbons close to oxygen, C-5 and C-6, displayed a peak at 52.7 ppm. Finally, peaks at 46.1 and 47.1 ppm accounted for the sp3 carbons, C-1 and C-4, and C-9. Impurities of ethyl acetate appeared at 46.6, 25.8, and 21.0 ppm accompanied with acetone at 30.9 ppm.
The IR spectrum revealed a peak at 2982 cm-1 representing the C-H stretches. A peak at 1840 cm-1 accounted for the carbonyl functional group, while a peak at 1767 cm-1 accounted for the alkene bond. A peak at 1089 cm-1 represented the carbon-oxygen functional group.
In order to distinguish between the two possible isomers, properties such as melting point and spectroscopy data were analyzed. The exo product possessed a melting point in the range of 140-145 °C which is significantly lower than the endo product. The observed melting point in this experiment supported the production of the endo isomer. The 1H NMR spectum exhibited a doublet of doublets at 3.57 ppm for the endo isomer. The exo isomer would possess a triplet around 3.50 ppm due to the difference in dihedral angle between the hydrogen molecules of H-1 and H-4, and H-5 and H-6 (Figure 1). A peak at 3.00 ppm would appear in the exo isomer spectra as opposed to a peak at 3.60 ppm as shown in the observed endo product.3 This is because of the interaction and coupling with the H-5 and H-6, as displayed in Figure 1.
Conclusion Through the Diels-Alder reaction, 27.6% yield of cis-Norbornene-5,6-endo-dicarboxylic anhydride was produced. The distinction of the presence of the endo isomer was proven by analyzing physical properties of both possible isomers.
Experimental
General: All reagents were provided by Sigma-Aldrich from Texas A&M University Chemistry Department. 1H and 13C spectra were taken on a Mercury 300 MHz NMR spectrometer. An IR spectrum was recorded on PerkinElmer UATR Two Spectrophotometer.
Cis-Norbornene-5,6-endo-dicarboxylic anhydride Cyclopentadiene was previously prepared through the cracking of dicyclopentadiene and kept under cold conditions. In a 25 mL Erlenmeyer flask, maleic anhydride (1.02 g, 10.4 mmol) and ethyl acetate (4.0 mL) were combined, swirled, and slightly heated until completely dissolved. To the mixture, ligroin (4 mL) was added and mixed thoroughly until dissolved. Finally, cyclopentadiene (1 mL, 11.9 mmol) was added to the mixture and mixed extensively. The reaction was cooled to room temperature and placed into an ice bath until crystallized. The crystals were isolated through filtration in a Hirsch funnel. The product had the following properties: 0.47 g (27.6% yield) mp: 163-164 °C (lit: 164 °C). 1H NMR (CDCl3, 300 MHz) δ: 6.30 (dd, J=1.8 Hz, 2H), 3.57 (dd, J=7.0 Hz, 2H), 3.45 (m, 2H), 1.78 (dt, J=9.0,1.8 Hz, 1H), 1.59 (m, 1H) ppm. 13C NMR (CDCl3, 75Hz) δ: 171.3, 135.5, 52.7, 47.1, 46.1 ppm. IR 2982 (m), 1840 (s), 1767 (s), 1089 (m) cm-1.
Supporting information IR, 1H NMR and 13C NMR spectra of cis-norborene-5,6-endo-dicarboxylic anhydride are attached.
1 Martin, J.; Hill, R.; Chem Rev, 1961, 61, 537-562.
2 Pavia, L; Lampman, G; Kriz, G; Engel, R. A Small Scale Approach to Organic Laboratory Techniques, 2011, 400-409.
3 Myers, K.; Rosark, J. Diels-Alder Synthesis, 2004, 259-265.
