Regeneration Of Planaria Dugesia and Annelid Lumbriculus
Abstract
Planaria Dugesia and Annelid Lumbriculus were observed in three different solutions (freshwater, 2.5mM locostatin, and 2.5 mM colchicine) after being cut in half sagittally. The purpose of this experiment was to test the significance of the worms’ environment on their regeneration process. Over the course of ten days, all worms in colchicine eventually died while both species of worms in freshwater and in locostatin solution survived and successfully completed regeneration. The results are significant because it exemplifies that regeneration can not occur in the presence of colchicine which impedes mitosis, but the process of regeneration is not disturbed by locostatin, which is an anti-migratory chemical.
Introduction
Previous studies have been conducted on Planarian and Annelid regeneration. One such experiment that relates to our regeneration experiment concluded that “regeneration in planarians involves the production of new tissue at the wound surface through cell proliferation” (Reddien, Alvarado 2004). Additional studies also conclude that “the regeneration processes in planarians and annelids usually found that an indifferent type of cell, variously termed formative cell, regeneration cell, or neoblast, plays an important role” (Hyman, 1940). These conclusions provide information to form a hypothesis in our experiment regarding the differing solutions that are exposed to.
Methods
- Obtain six Petri dishes. Label each one according to the solution it will hold.
- Fill two dishes with fresh water. This will serve as the control.
- This control exists to demonstrate the process of regeneration in Planaria and Annelid’s natural environment. Nothing in this solution will impede mitosis or migration in the worms, and results of the control will serve as a comparison to the observations obtained from the independent variable.
- Dilute both colchicine and locostatin solutions to 2.5 mM.
- Colchicine is used because it prevents mitosis from occurring by inhibiting microtubule reactions by binding to tubulin.
- Locostatin is used because it disrupts the function of proteins that are essential for migratory cell movement.
- Fill two dishes with colchicine and the remaining two dishes with locostatin.
- Obtain fifteen Planaria and fifteen Annelids.
- Put five Planaria in each of the three labeled Planaria Petri dishes, put five Annelids in each of the three Annelid Petri dishes.
- Use a razor blade to make a sagittal cut in each worm, making two equal length halves of the worm.
- Leave worms in solutions for ten days, refreshing the solutions in each dish every two days.
- Observe the changes in the worms every day for the first five days and then every other day until day ten.
- Record results and photograph regenerated worms.
Results
|
Solution |
Day |
Planaria |
Annelids |
| Locostatin | 1 | 10/10 half sized worms alive and moving | 10/10 half sized worms alive and moving |
| Colchicine
Control |
10/10 half sized worms alive and moving10/10 half sized worms alive and moving | 10/10 half sized worms alive and moving10/10 half sized worms alive and moving | |
| Locostatin | 2 | 3 active head pieces, 2 elongated head pieces, 5 inactive tails, still undergoing regeneration | 8 active long pieces, 2 active short segments, seem farther along in regeneration than other Annelids |
| Colchicine
Control |
4 inactive tail segments 1 active head only, most seemed dead, regeneration uncertain
5 active, mostly fully formed, 5 inactive tail segments |
4 active long, 7 active short, seemed very healthy, even more so than control, still in early stages of regeneration
7 active long segments, 2 active shor, 1 active long, seemed healthy, proper regeneration still in early stages |
|
| Locostatin | 3 | some regeneration visible, anterior segments motile, posterior segments starting to have visible eye spot formation, but no elongation or tapering. Approximately 2/3 starting sample size was in the dish, not accounting for loss of organisms. | less motile organisms, some regeneration at cut points, however this could have been simple wound closure as the lighter colored areas were significantly smaller than control. Anterior/posterior mouthpart differentiation visible. |
| Colchicine
Control |
complete death and necrotization, the rot filled water was discarded. Experimental setup terminated here.regeneration visible. Anterior segments with proper morphology, and upon attempts to capture, were motile. No response from the posterior segments, eyespot formation visible in some organisms | upon stimulation motile, no visible regeneration
highly motile organisms, visible regeneration as lighter color band at one end. No visible mouthparts on the posterior segments. Anterior segments significantly more active |
|
| Locostatin | 4 | anterior sections defined and motile, posterior sections remain amotile. All have visible eyespots, however lack a defined head section. | still less motile overall than controls, more clearly defined regenerative zone, posterior segments still far less motile than anterior. |
| Colchicine
Control |
not present (omit from subsequent observations)
motile completely regenerated organisms for anterior sectioning. Some posterior sections unchanged, suspect that these are either dead or extremely slow in developing. By the numbers at least one posterior section has developed into a motile organism with a defined head. |
no movement without stimulation, no visible regenerationhighly motile organisms with essentially conserved morphology. Some mouthpart formation visible on posterior sections. Anterior sections completely recovered. Regeneration zone clearly visible. Posterior section mouthpart formation appears to occur opposite the sight of the cut | |
| Locostatin | 5 | 3 active that are missing tails, 1 half formed, 6 inactive pieces, seem alive but regeneration not complete, may be stalled or done | 7 active long, 2 active short, seem healthy, tails are pointy, needle-like |
| Colchicine
Control |
4 active complete Planaria, 6 incomplete segments, regeneration seems to have occurred normally |
7 active long, 1 active half size, 1 inactive small
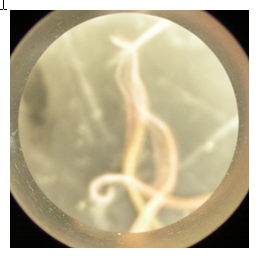
8 almost fully formed, 2 semi-active short segments, seem healthy but tails are stubby (Figure 1) |
|
| Locostatin | 6 | 8/10 living, 1/10 dead and intact but very small, 1/10 degraded bits floating around | 10/10 alive and original size |
| Control | 10/10 living, 8/10 are half the size of the original, 2/10 are very small (quarter of the size of original) | 8/10 alive and original size, 1/10 dead and half size, 1/10 degraded bits | |
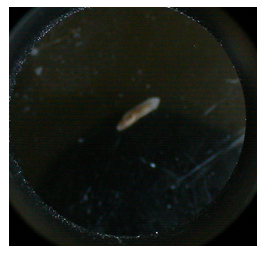
| Locostatin | 8 | 6 active with stubby tails, very sluggish (Figure 5) | 9 active full one active short, pointed tails (Figure 2) |
| Colchicine
Control |
5 full, active; 1 inactive full; 1 half sized active, 1 inactive half |
not present (omit from subsequent observations)
8/10 alive and original size, 1/10 dead and half size, 1/10 degraded bits |
|
| Locostatin | 10 | 8/10 living, 1/10 dead and intact but very small, 1/10 degraded bits floating around (Figure 3) | 8/10 alive, 2/10 dead and degraded(Figure 4) |
| Control | 9/10 alive, 8/10 normal size and 1/10 half sized 1/10 was two dead halves | 10/10 alive and original size, active without needing stimulation |
Conclusions
It can be concluded that the regeneration process of Planaria and Annelids in a solution of colchicine is impeded and eventually leads to the death and degeneration of both types of worm. Due to observations in this experiment, Planaria and Annelids in solutions of locostatin regenerated completely, with no significant differences from the results of the control plates. Drawing from observations from all six Petri dishes throughout the ten days, it is accurate to say that regeneration is able to occur in a freshwater environment and a diluted locostatin environment. Furthermore, as observed, if the worms cannot regenerate properly, like in the colchicine environment, they will eventually die. These findings support our original hypothesis that stated that both types of worms require the process of mitosis to properly regenerate. This hypothesis was also supported by findings from Hyman’s experiment that concluded “In regeneration of [worms], Faulkner (1932) reported that the neoblasts multiply and differentiate into all the new parts of the regenerate.” This conclusion informed us that cell multiplication was essential for regeneration to occur. Since neither the Planaria nor the Annelids were observed histologically at high magnification, we did not conclude that all cell types divided and differentiated into their correct locations. Had we observed at a higher level of magnification, we could have confirmed or denied the correct location of various cell types.
Literature Cited
Hymann, Libbie H., “Aspects of Regeneration in Annelids” The American Naturalist Vol. 74, No. 755 (Nov. – Dec., 1940), pp. 513-527
Published by: The University of Chicago Press for The American Society of Naturalists
Reddien, Peter W., and Alejandro Sanchez Alvarado. “Fundamentals Of Planarian Regeneration.” Annual Review of Cell and Developmental Biology 20.1 (2004): 725-57. Print.